Popular on TelAve
- Ice Melts. Infrastructure Fails. What Happens to Clean Water?
- Amicly Launches as a Safety-First Social App Designed to Help People Build Real, Meaningful Friendships
- The Legal AI Showdown: Westlaw, Lexis, ChatGPT… or EvenSteven?
- François Arnaud, star of Heated Rivalry, is the real-life inspiration behind Christopher Stoddard's novel At Night Only
- Purple Heart Recipient Honored by Hall of Fame Son In Viral Tribute Sparking National Conversation on Service Fatherhood, Healing and Legacy
- Off The Hook YS (NY SE: OTH) Executes Transformational Apex Acquisition, Creating Vertically Integrated Marine Powerhouse with $60M Inventory Capacity
- AOW Event Sponsored By The Stanglwirt Resort a renowned five-star Austrian wellness destination
- Special Alert: Undervalued Opportunity: IQSTEL (N A S D A Q: IQST) Positioned for Explosive Multi-Year Growth
- Triple-Digit Growth, Strategic N A S D A Q Uplist, Plus A Scalable Healthcare Rollout Model: Stock Symbol: CDIX
- ClearBeam Networks Launches HomeStation: Home Phone 2.0
Similar on TelAve
- Why Your Berberine Failed: RevGenetics Unveils the Absorption Gap Solution
- Jackets for Jobs Hosts Smart & Sexy® Day Detroit for Women's History Month
- $IBG accelerates toward transformative merger with BlockFuel as $6 million raise fuels dual-industry growth strategy: N A S D A Q: IBG
- High-Growth Power Infrastructure Play Targets AI Boom: 1606 Corp. Executes Aggressive Texas Expansion Strategy: 1606 Corp. (Stock Symbol: CBDW) $CBDW
- Accelerating the Transformation into a U.S. Nuclear Fuel Cycle Leader: Frontier Nuclear and Minerals Inc. (N A S D A Q: FNUC)
- Ozz Metals Ltd Secures 1-Tonne Gold Offtake Agreement
- Compliant Workspace announces partnership with Blackpoint Cyber
- Event Solutions Enters New Era: Announces New Leadership
- Scoop Social Co. Launches a New Era of Mobile Hospitality — One Truck, Two Experiences
- Record Sales Growth After Strategic Acquisitions; New Distribution Agreements for Established Premium Cigar Supplier: Green Leaf Innovations $GRLF
Acquisition of Clinics Generating $100 Million to Complement Progress on Accelerated Drug Approval for Bipolar Depression NRx Pharmaceuticals: NRXP
TelAve News/10838656
NRXP: Potential Acquisition and Financing Agreements for $30 Million in Currently-Operating Interventional Psychiatry Clinics.
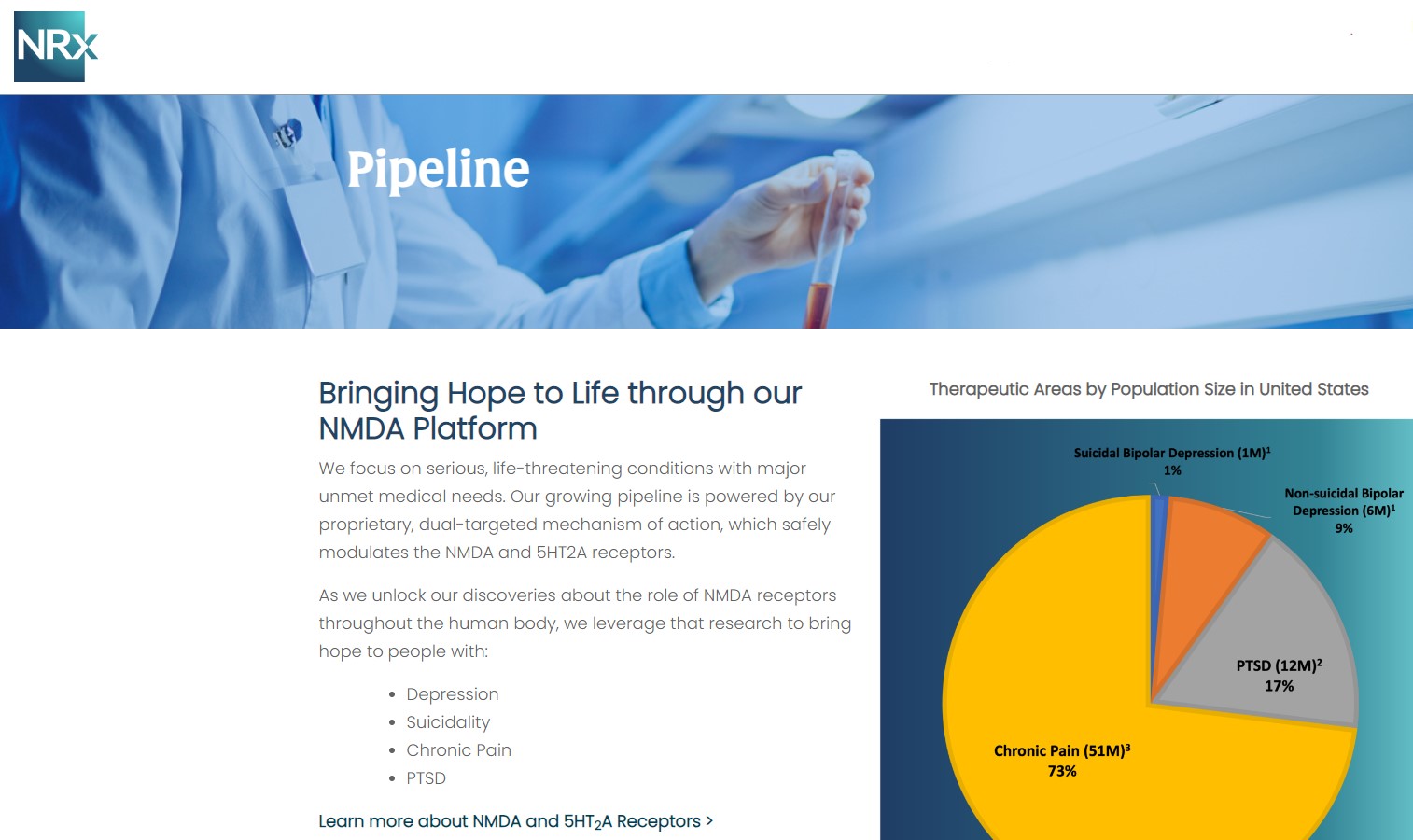
MIAMI - TelAve -- NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Potential Acquisition and Financing Agreements for $30 Million in Currently-Operating Interventional Psychiatry Clinics
On August 26th NRXP announced signing a non-binding Term Sheet for non-dilutive, nonconvertible debt acquisition funding of its first interventional psychiatry clinics (ketamine clinic acquisitions), together with the signing of a Term Sheet for five currently operational clinics in the Western United States. In addition to the currently-signed non-binding Term Sheet, NRXP has been offered non-binding lending commitments it believes are sufficient to assemble/acquire a network of operational clinics with revenues in excess of $100 million. NRXP anticipates potential operations in the United States, France, and the United Kingdom.
More on TelAve News
This non-dilutive acquisition funding is in addition to the over $60 million in potential equity funding previously offered upon public listing of HOPE Therapeutics shares on a public exchange. Additional information to be presented by NRXP at upcoming HC Wainwright Annual Global Investment Conference in New York, September 9-11, 2024.
NRXP has formed partnerships to license clinical trial data from a French Government-funded trial and two NIH-funded trials all of which demonstrate efficacy of racemic Intravenous ketamine against depression and two of which demonstrate statistically significant benefit vs suicidality.
In March 2024 NRXP demonstrated the formulation of a pH neutral patentable form of IV ketamine that it anticipates will have widespread applicability both in treatment of depression and chronic pain.
NRXP presented final data from the recently completed phase 2b/3 trial of NRX-101 in suicidal bipolar depression4 at the American Society of Clinical Psychopharmacology's annual meeting. These data demonstrated a significantly improved safety profile versus the standard of care, as demonstrated by a clinically significant reduction in akathisia (P=0.025) and time to sustained remission from suicidality (P=0.05). Akathisia is an adverse event seen with antidepressant medications considered by many experts to be a precursor to suicide. Given the vital need for safer medications in this at-risk population, we plan to submit an NDA for Accelerated Approval to the US FDA for treatment of bipolar depression patients with suicidality or akathisia, based on these data as well as additional data from our STABIL-B5 trial.
HOPE Therapeutics, is an NXRP subsidiary that will focus on the delivery of advance psychiatric treatments, including ketamine-focused treatment for depression and suicidality. Unlike the core business of NRXP, that is focused on biotechnology Research and Development, HOPE is organized around consolidating existing best-in-class clinics into a nationwide network. This has been done previously and without much success with clinics that are not necessarily psychiatrist-led. With Hope ownership as an asset of NRXP, this will further strengthen the NRXP balance sheet and aims to further enhance shareholder value.
More on TelAve News
NRX-101 for Treatment of Chronic Pain:
In 2023, NRXP licensed US Patent 8,653,120 for the use of DCS in chronic pain and filed a now-accepted Investigational New Drug (IND) application with the FDA to initiate commercial drug development of NRX-101 in chronic pain. Data lock has now been achieved in a 200-person randomized prospective trial funded by the US DOD.
During Q3 2023, NRXP tested NRX-101 and its components against resistant pathogens that appear on the Congressionally mandated Qualified Infectious Disease Product (QIDP) list and proved in vitro effectiveness against antibiotic-resistant E. coli, Pseudomonas, and Acinetobacter. Accordingly, NRXP was granted QIDP designation, Fast Track Designation, and Priority Review by the US FDA in January 2024.
Should NRX-101 succeed in clinical trials, NRXP will consider developing a follow-on product that is anticipated to achieve another 20 years of patent exclusivity.
NRXP recently announced data demonstrating that NRX-101 does not compromise the intestinal microbiome, unlike common antibiotics including Clindamycin and Ciprofloxacin. Should these findings be documented in human patients, NRX-101 would represent the only treatment for cUTI that does not cause C. Difficile infection.
Additionally, should NRXP or its partners succeed in serving 10% of the cUTI market, NRXP believes that the revenue from NRX-101 has the potential to be hundreds of million annually, based on 3 million cases per year in the US and potential pricing of over $3,500/course of therapy.
Media Contact
Company Name: NRx Pharmaceuticals, Inc.
Contact Person: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Country: United States
Website: https://www.nrxpharma.com/
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Potential Acquisition and Financing Agreements for $30 Million in Currently-Operating Interventional Psychiatry Clinics
On August 26th NRXP announced signing a non-binding Term Sheet for non-dilutive, nonconvertible debt acquisition funding of its first interventional psychiatry clinics (ketamine clinic acquisitions), together with the signing of a Term Sheet for five currently operational clinics in the Western United States. In addition to the currently-signed non-binding Term Sheet, NRXP has been offered non-binding lending commitments it believes are sufficient to assemble/acquire a network of operational clinics with revenues in excess of $100 million. NRXP anticipates potential operations in the United States, France, and the United Kingdom.
More on TelAve News
- Tint Academy Training in Dallas Texas: Learn Window Tint & PPF Installation
- ASTPP Launches Advanced Telecom Billing Software to Simplify Billing for VoIP Providers
- $IBG accelerates toward transformative merger with BlockFuel as $6 million raise fuels dual-industry growth strategy: N A S D A Q: IBG
- High-Growth Power Infrastructure Play Targets AI Boom: 1606 Corp. Executes Aggressive Texas Expansion Strategy: 1606 Corp. (Stock Symbol: CBDW) $CBDW
- Accelerating the Transformation into a U.S. Nuclear Fuel Cycle Leader: Frontier Nuclear and Minerals Inc. (N A S D A Q: FNUC)
This non-dilutive acquisition funding is in addition to the over $60 million in potential equity funding previously offered upon public listing of HOPE Therapeutics shares on a public exchange. Additional information to be presented by NRXP at upcoming HC Wainwright Annual Global Investment Conference in New York, September 9-11, 2024.
NRXP has formed partnerships to license clinical trial data from a French Government-funded trial and two NIH-funded trials all of which demonstrate efficacy of racemic Intravenous ketamine against depression and two of which demonstrate statistically significant benefit vs suicidality.
In March 2024 NRXP demonstrated the formulation of a pH neutral patentable form of IV ketamine that it anticipates will have widespread applicability both in treatment of depression and chronic pain.
NRXP presented final data from the recently completed phase 2b/3 trial of NRX-101 in suicidal bipolar depression4 at the American Society of Clinical Psychopharmacology's annual meeting. These data demonstrated a significantly improved safety profile versus the standard of care, as demonstrated by a clinically significant reduction in akathisia (P=0.025) and time to sustained remission from suicidality (P=0.05). Akathisia is an adverse event seen with antidepressant medications considered by many experts to be a precursor to suicide. Given the vital need for safer medications in this at-risk population, we plan to submit an NDA for Accelerated Approval to the US FDA for treatment of bipolar depression patients with suicidality or akathisia, based on these data as well as additional data from our STABIL-B5 trial.
HOPE Therapeutics, is an NXRP subsidiary that will focus on the delivery of advance psychiatric treatments, including ketamine-focused treatment for depression and suicidality. Unlike the core business of NRXP, that is focused on biotechnology Research and Development, HOPE is organized around consolidating existing best-in-class clinics into a nationwide network. This has been done previously and without much success with clinics that are not necessarily psychiatrist-led. With Hope ownership as an asset of NRXP, this will further strengthen the NRXP balance sheet and aims to further enhance shareholder value.
More on TelAve News
- Ozz Metals Ltd Secures 1-Tonne Gold Offtake Agreement
- Jet Set: The Ultimate Coachella Afterparty
- Heritage at Manalapan Introduces New Single-Family Home Community in One of Monmouth County's Most Desirable Locations
- Compliant Workspace announces partnership with Blackpoint Cyber
- Crossover Named Official Distributor of Meshmerize Dynamic Mesh Networking Solutions
NRX-101 for Treatment of Chronic Pain:
In 2023, NRXP licensed US Patent 8,653,120 for the use of DCS in chronic pain and filed a now-accepted Investigational New Drug (IND) application with the FDA to initiate commercial drug development of NRX-101 in chronic pain. Data lock has now been achieved in a 200-person randomized prospective trial funded by the US DOD.
During Q3 2023, NRXP tested NRX-101 and its components against resistant pathogens that appear on the Congressionally mandated Qualified Infectious Disease Product (QIDP) list and proved in vitro effectiveness against antibiotic-resistant E. coli, Pseudomonas, and Acinetobacter. Accordingly, NRXP was granted QIDP designation, Fast Track Designation, and Priority Review by the US FDA in January 2024.
Should NRX-101 succeed in clinical trials, NRXP will consider developing a follow-on product that is anticipated to achieve another 20 years of patent exclusivity.
NRXP recently announced data demonstrating that NRX-101 does not compromise the intestinal microbiome, unlike common antibiotics including Clindamycin and Ciprofloxacin. Should these findings be documented in human patients, NRX-101 would represent the only treatment for cUTI that does not cause C. Difficile infection.
Additionally, should NRXP or its partners succeed in serving 10% of the cUTI market, NRXP believes that the revenue from NRX-101 has the potential to be hundreds of million annually, based on 3 million cases per year in the US and potential pricing of over $3,500/course of therapy.
Media Contact
Company Name: NRx Pharmaceuticals, Inc.
Contact Person: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Country: United States
Website: https://www.nrxpharma.com/
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: Corporate Ads
0 Comments
Latest on TelAve News
- Greg Wier Announces the Release of More Than Just Luck
- Nieuwe standaard in webdesign: Professionele website laten maken voor het MKB vanaf €249 door Websitepioniers
- Conexwest: Shipping Containers Are Powering the Next Generation of Bitcoin Mining Infrastructure
- Hypnotherapy Finder Announces Official US Wide Launch After Successful Soft Launch
- Melzi Job Coach Launches on iOS and Android: A Privacy-First AI Career Engine Built for Execution
- Training Lofts Launches $1,099 Unlimited Training Membership Featuring Semi-Private Coaching, Nutrition Support, and Recovery Services
- American Properties Realty, Inc. Leadership Attends NAHB International Builders' Show in Florida
- $317M Revenue and a Clear Path to $1B: $IQST is Positioned for a Major Profitability Inflection
- ASI Hosts 2026 Executive Business Summit for Global Partner Community
- Pastor Saeed Abedini Releases THE TRUTH – Volume 1, A Deeply Personal Story of Faith, Struggle, and Redemption
- New Book Warring From the Standpoint of the Throne Room Calls Believers to Pray From Victory
- Scotch Whisky Market Dislocation Creates Compelling Entry Opportunity for Long-Term Investors
- Peccioli Becomes New Orleans: In July 2026, the magic of jazz comes to Tuscany
- $6 Million Funding Secured as Retail Expansion, Operational Streamlining, and Asset-Light Strategy Position the Company for Accelerated Growth $SOWG
- The "Unsexy" Business Quietly Creating 130+ New Entrepreneurs Across America — From Alaska to Puerto Rico
- Veteran Launches GTG Energy: Nicotine-Free Pouch as Americans Rethink Addiction, Focus, and What Fuels Performance
- RecallSentry™ App Launch — Your Home Safety Hub — Free on iOS & Android
- Award-Winning Director Crystal J. Huang's Under-$50K Film "The Ritual House" Wins Best Horror Feature at Golden State Film Festival
- Grads aren't getting hired — here's what we're doing about it
- K2 Integrity Enhances Technology Capabilities Through Acquisition of Leviathan Security Group