Popular on TelAve
- Attorney Joseph C. Kreps Files Lawsuit to Stop Alabama State Board of Pharmacy's Unlawful "Revenue-First" Rulemaking - 119
- Advantage Marketing Launches 3-Minute Assessment to Help SMBs Diagnose and Fix Marketing Gaps - 103
- Card makers turn to Pink and Main for tools to support their craft
- Evermore Bliss Launches AI Wedding Speech Writer to Help Users Create Personalized, Heartfelt Toasts
- Mensa Brings National Board Game Competition to Northern Virginia April 16-19
- Acuvance Appoints Sandeep Sabharwal to Board of Directors, Strengthening Leadership to Support Continued Platform Growth
- Instant IP Launches Rapid Takedown Service to Combat IP Theft, Deepfakes, and Copycat Websites
- Special Alert! Highly Undervalued Stock: $317M Revenue in 2025 for Telecom Leader IQSTEL, Inc. (N A S D A Q: IQST)
- As Global Tensions Rise, Demand Grows for Private Spaces to Process Thoughts and Speak Freely Online
- Mac Mountain Selects netElastic vRouter for LightCraft Broadband-as-a-Service Platform
NRx Pharmaceuticals (N A S D A Q: NRXP) Accelerates Into National Spotlight as Manufacturing Launch, Federal Policy & AI-Driven Breakthroughs Converge
TelAve News/10895229
Commercial-Ready Ketamine Production, FDA Alignment, and Expanding AI Neurotherapy Platform Position $NRXP at the Center of a Multi-Billion-Dollar Mental Health Transformation
MIAMI - TelAve -- NRx Pharmaceuticals (N A S D A Q: NRXP) is rapidly advancing beyond the traditional biotech model, emerging as a commercially prepared, AI-enabled mental health platform at a time when federal policy, regulatory clarity, and technological innovation are converging in its favor.
The company's trajectory has already been defined by a rare alignment of catalysts—most notably the April 18, 2026 Presidential Executive Order accelerating approval of breakthrough therapies for serious mental illness. This directive is expected to expand the use of real-world evidence, compress regulatory timelines, and introduce priority mechanisms that directly benefit companies like NRx, positioning it within a policy-supported pathway toward faster approvals and broader adoption.
Commercial Inflection Point: Manufacturing Now Underway
Layered on top of this macro tailwind, NRx has taken a decisive operational step forward with the initiation of its first commercial-scale manufacturing order for preservative-free ketamine—positioning the company ahead of anticipated FDA approval under its ANDA pathway expected in Summer 2026.
This marks a clear shift from development to execution.
Key manufacturing advantages include:
NRx's formulation also eliminates Benzethonium Chloride, a preservative no longer permitted in new drugs, creating a modern, differentiated product profile aligned with evolving regulatory standards.
Federal Policy Alignment Creates a Powerful Tailwind
The Presidential directive to accelerate psychedelic and breakthrough therapies is more than a headline—it represents a structural shift in how mental health treatments are approved and deployed.
More on TelAve News
NRx is already aligned with this framework, positioning itself to benefit from:
This transforms the company's regulatory pathway into one that is increasingly policy-supported and time-efficient.
FDA Alignment Anchors the Investment Thesis
At the core of the story is a rare regulatory breakthrough.
NRx has secured agreement from the FDA to proceed with its New Drug Application for NRX-100 (preservative-free ketamine) using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This enables a targeted NDA submission as early as June 2026 while expanding the indication to include treatment-resistant depression with suicidality.
Regulatory positioning highlights:
Dual FDA Pathways Create Layered Upside
NRx is uniquely positioned with two concurrent regulatory opportunities.
While the NDA pathway represents long-term value creation, the ANDA pathway—now supported by active manufacturing—introduces near-term commercialization potential.
Dual catalyst structure:
This parallel progression is rare and significantly enhances visibility for investors.
From Biotech to Platform: AI + Neurotechnology Expansion
NRx is no longer a single-product company—it is building an integrated platform combining pharmaceuticals, neurostimulation, and artificial intelligence.
Through its NRx Defense Systems subsidiary, the company is advancing robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy, targeting military and first responder populations.
At the same time, AI integration is transforming care delivery.
Platform expansion includes:
Commercial Infrastructure Already Generating Revenue
More on TelAve News
NRx is not waiting for approval to build its business.
Through HOPE Therapeutics, the company has established a growing network of interventional psychiatry clinics delivering ketamine therapy, TMS, and neuroplastic treatments.
These clinics are:
This creates a ready-made commercialization engine.
Financial Position Supports Execution
NRx has strengthened its balance sheet through reduced operating expenses, elimination of debt, and sufficient capital to support operations through 2026.
This provides the flexibility to execute across manufacturing, regulatory, and commercialization initiatives simultaneously.
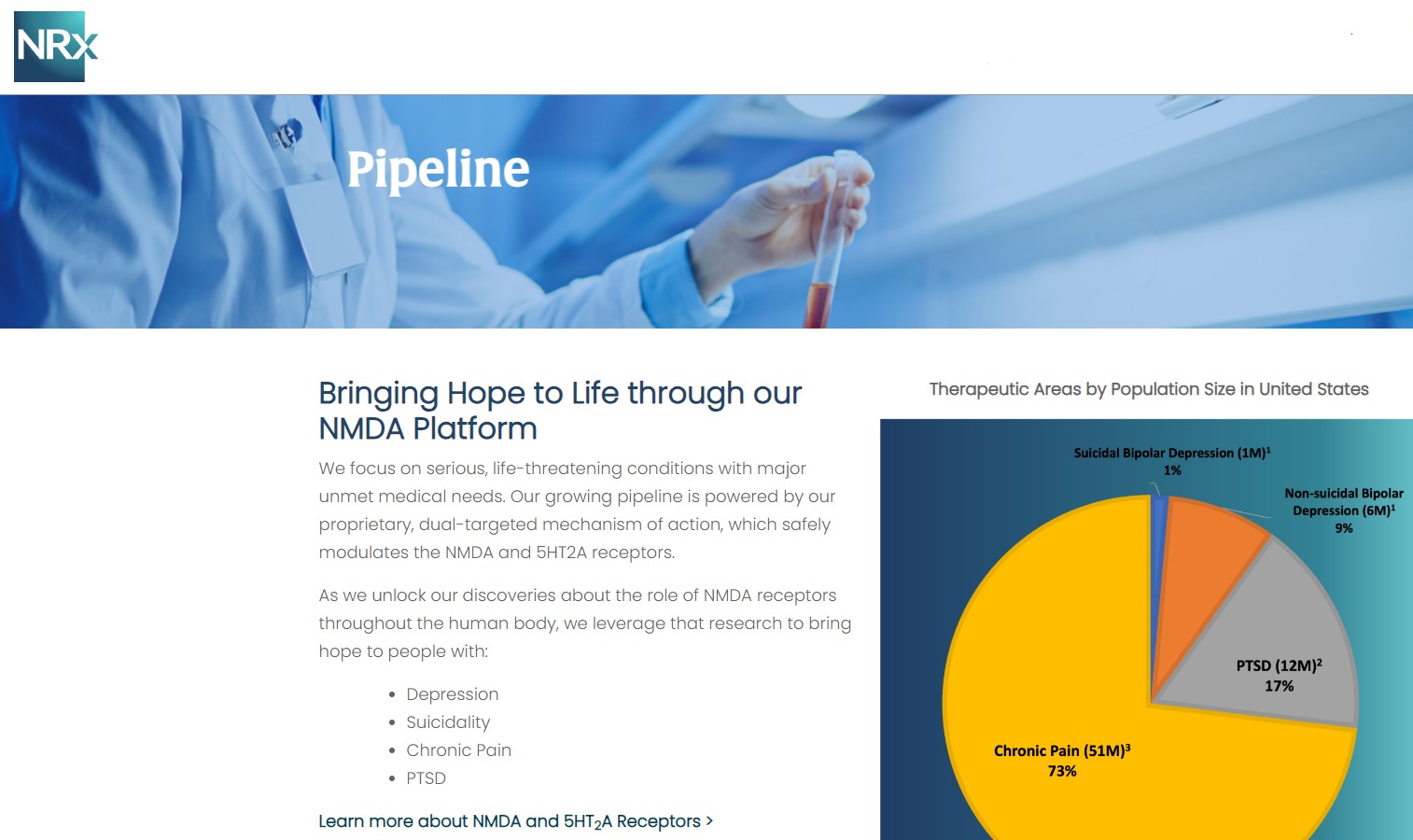
A Massive, Urgent Market Opportunity
NRx is targeting one of the most critical unmet needs in healthcare.
With ketamine shortages persisting and demand for rapid-acting therapies accelerating, $NRXP is entering the market at a time of both urgency and opportunity.
Final Take: From Catalyst Story to Execution Story
NRx Pharmaceuticals is no longer simply approaching catalysts—it is activating them across multiple fronts simultaneously.
With:
$NRXP represents a rare convergence of timing, technology, and execution.
This is no longer a company waiting for approval—
It is a company preparing to deliver at scale.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
The company's trajectory has already been defined by a rare alignment of catalysts—most notably the April 18, 2026 Presidential Executive Order accelerating approval of breakthrough therapies for serious mental illness. This directive is expected to expand the use of real-world evidence, compress regulatory timelines, and introduce priority mechanisms that directly benefit companies like NRx, positioning it within a policy-supported pathway toward faster approvals and broader adoption.
Commercial Inflection Point: Manufacturing Now Underway
Layered on top of this macro tailwind, NRx has taken a decisive operational step forward with the initiation of its first commercial-scale manufacturing order for preservative-free ketamine—positioning the company ahead of anticipated FDA approval under its ANDA pathway expected in Summer 2026.
This marks a clear shift from development to execution.
Key manufacturing advantages include:
- Completed GMP audits and validated registration batches
- Initial capacity designed to support meaningful first-year revenue
- Rapid scalability via blow-fill-seal process (10x traditional output)
- U.S.-based production addressing ongoing ketamine supply shortages
NRx's formulation also eliminates Benzethonium Chloride, a preservative no longer permitted in new drugs, creating a modern, differentiated product profile aligned with evolving regulatory standards.
Federal Policy Alignment Creates a Powerful Tailwind
The Presidential directive to accelerate psychedelic and breakthrough therapies is more than a headline—it represents a structural shift in how mental health treatments are approved and deployed.
More on TelAve News
- Michael H. Kaplan, Colorado Workers' Compensation Attorney, Rallies Athlete Unions Against Proposed Legislative "Carve-Outs"
- Viasat, Galaxy 1 Communications and L2 Aviation to bring avionics integration to Advanced Air Mobility
- Fulton County DA Fani Willis Officially Endorses Dr. Heavenly Kimes + Black Economic Agenda
- Bellwether Farm Presents Kerry Hill Lamb to His Majesty King Charles III During Historic U.S. State Visit
- New Study Finds Americans Judge Vacations on Value, Not Price — Signaling a Permanent Shift in How Travel Gets Booked
NRx is already aligned with this framework, positioning itself to benefit from:
- Expanded acceptance of real-world evidence
- Faster FDA review timelines
- Potential access to priority voucher programs
This transforms the company's regulatory pathway into one that is increasingly policy-supported and time-efficient.
FDA Alignment Anchors the Investment Thesis
At the core of the story is a rare regulatory breakthrough.
NRx has secured agreement from the FDA to proceed with its New Drug Application for NRX-100 (preservative-free ketamine) using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This enables a targeted NDA submission as early as June 2026 while expanding the indication to include treatment-resistant depression with suicidality.
Regulatory positioning highlights:
- No additional trials required
- Fast Track designation already granted
- 70,000+ patient real-world dataset supporting efficacy
- Expanded label strategy increasing addressable market
Dual FDA Pathways Create Layered Upside
NRx is uniquely positioned with two concurrent regulatory opportunities.
While the NDA pathway represents long-term value creation, the ANDA pathway—now supported by active manufacturing—introduces near-term commercialization potential.
Dual catalyst structure:
- ANDA approval (anticipated Summer 2026) → near-term revenue
- NDA submission (target June 2026) → transformational upside
This parallel progression is rare and significantly enhances visibility for investors.
From Biotech to Platform: AI + Neurotechnology Expansion
NRx is no longer a single-product company—it is building an integrated platform combining pharmaceuticals, neurostimulation, and artificial intelligence.
Through its NRx Defense Systems subsidiary, the company is advancing robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy, targeting military and first responder populations.
At the same time, AI integration is transforming care delivery.
Platform expansion includes:
- AI-driven patient monitoring ("Depression Thermometer")
- Real-time treatment optimization
- Robotic, precision-guided neurostimulation systems
- Potential non-dilutive government and defense funding channels
Commercial Infrastructure Already Generating Revenue
More on TelAve News
- Pomona Organic Launches New Website, Surpasses 10 Million Bottles Sold, and Opens Affiliate Program to Creators
- Postmortem Pathology Opens Sacramento Office Offering Private Autopsies for Families and Healthcare Investigations
- Postmortem Pathology, a leading provider of private autopsies, has announced its expansion into the Las Vegas market
- Kick'em Out Quick® Evictions Announces a New Endorsed Eviction Attorney in Atlanta / Fulton County, GA
- Crunchbase Ranks Phinge CEO #1 Globally: Meet Him At "Phinge Unveil", Preview Netverse Patented App-less Platform, Hardware & Netverse Intelligence NI
NRx is not waiting for approval to build its business.
Through HOPE Therapeutics, the company has established a growing network of interventional psychiatry clinics delivering ketamine therapy, TMS, and neuroplastic treatments.
These clinics are:
- Operational and revenue-generating
- Supported by government programs, insurers, and private pay
- Positioned for rapid scale post-approval
This creates a ready-made commercialization engine.
Financial Position Supports Execution
NRx has strengthened its balance sheet through reduced operating expenses, elimination of debt, and sufficient capital to support operations through 2026.
This provides the flexibility to execute across manufacturing, regulatory, and commercialization initiatives simultaneously.
A Massive, Urgent Market Opportunity
NRx is targeting one of the most critical unmet needs in healthcare.
- Over 13 million Americans seriously consider suicide annually
- Rising global rates of treatment-resistant depression and PTSD
With ketamine shortages persisting and demand for rapid-acting therapies accelerating, $NRXP is entering the market at a time of both urgency and opportunity.
Final Take: From Catalyst Story to Execution Story
NRx Pharmaceuticals is no longer simply approaching catalysts—it is activating them across multiple fronts simultaneously.
With:
- Commercial manufacturing now underway
- Federal policy accelerating approvals
- FDA-aligned pathways requiring no new trials
- Dual regulatory opportunities
- Expanding AI and neurotechnology platform
- Revenue-generating clinical infrastructure
$NRXP represents a rare convergence of timing, technology, and execution.
This is no longer a company waiting for approval—
It is a company preparing to deliver at scale.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on TelAve News
- Karen D. Gentry Announces New Book Focused on Relationships and Personal Growth
- New plusOne Research Finds the Orgasm Gap Is a 30-Point Chasm — and Confirms It Isn't Biology
- CCHR Report Links 145 Violent Incidents to Psychiatric Drug Exposure, Urges National Oversight and Action
- Food Journal Magazine Unveils Its Latest 'Best of Los Angeles' Culinary Discoveries
- Boston Industrial Solutions Launches Natron® 717S Series: A New Flexible UV LED Ink for Ricoh GH2220 Printheads
- 5 Things NYC Courier Services Won't Tell You About How Same-Day Delivery Actually Works
- Tropidelic Links Up With International Reggae Star Collie Buddz and Eli Mac for Feel-Good Breakthrough Single "Follow Your Nature"
- Save 15 Percent on Florida Keys Accommodations with KeysCaribbean's 'Advance Purchase Rate Discount'
- Atelier 411 Studios and Columbus Fashion Council Present Red Carpet Experience at Gateway Film Center for The Devil Wears Prada 2
- Solo Indie Developer Launches Not Pink, a Couples App Built Around a Four-Phase Planning Journey
- Hazel E Celebrates Birthday with Luxury "Goddess" Yacht Experience in Marina del Rey
- Joseph Neibich sits down with Bold Jounrey (aka Joseph Nybyk)
- AI Suite 360 Launches Done-For-You AI Implementation to Rescue SMBs from the "Frankenstein Tax"
- CX Network Releases Report on the Best AI Support Tools for SaaS Companies 2026
- Outlier Pest Season Hits Willamette Valley as Mild Winter Drives Early Surge in Ant and Rodent Activity
- Lokal Media House Wins Web Excellence Award for Black Plumbing Redesign
- Lick Expands Flavored Massage Oil Collection with 10 New Indulgent Cream-Inspired Scents
- Former Eatigo Co-Founder Michael Cluzel Joins SPARK To Reinvent Dating Through Real Experiences
- New Research Identifies "Vacation Compatibility Gap" as the Hidden Force Shrinking How Long and With Whom Americans Travel
- Melospeech Inc. Awarded New NYSDOH BEI Contract in New York