Popular on TelAve
- Amicly Launches as a Safety-First Social App Designed to Help People Build Real, Meaningful Friendships
- Purple Heart Recipient Honored by Hall of Fame Son In Viral Tribute Sparking National Conversation on Service Fatherhood, Healing and Legacy
- AOW Event Sponsored By The Stanglwirt Resort a renowned five-star Austrian wellness destination
- Triple-Digit Growth, Strategic N A S D A Q Uplist, Plus A Scalable Healthcare Rollout Model: Stock Symbol: CDIX
- Special Alert: Undervalued Opportunity: IQSTEL (N A S D A Q: IQST) Positioned for Explosive Multi-Year Growth
- Cancun All Inclusive is ready for Spring Break 2026 with new Resorts, Exclusive Deals, activities and more!
- ClearBeam Networks Launches HomeStation: Home Phone 2.0
- Best Book Publishing Company for Aspiring Authors
- Inkdnylon Earns BBB Accreditation for Verified Business Integrity
- Average US gambler spends $210 per month in 2026
Similar on TelAve
- Monexplora Explains the Options Mechanics Behind March's Tech Selloff and VIX Surge
- Compliance Alert: Maryland, Texas Regulate Use of Artificial Intelligence in Utilization Reviews
- EFA Announces 2026 Editorial Rate Chart
- Shoutout Joseph Neibich aka Nybyk
- AI Disruption Meets Marine Scale: Off The Hook YS, Inc. (N Y S E American: OTH) Targets Breakout Growth with NextBoat Launch and Aggressive Expansion
- Targeting the Billion-Dollar U.S. Countermeasure Market With AI-Driven Biodefense Platform: Lunai Bioworks (N A S D A Q: LNAI)
- New Global Standard for Transparency Across Critical Resources and Energy Markets: SMX (Security Matters) PLC (N A S D A Q: SMX)
- Forced Psychiatric Hospitalization Fails Vulnerable People: CCHR Urges Repeal Amid Rising U.S. Policies
- Surging Into High-Performance AI With $AMD Partnership, Patent Expansion, and Strengthened Balance Sheet: Avalon GloboCare Corp. (N A S D A Q: ALBT)
- NEW MANAGEMENT BOOK: Creating a Joy-Centric Culture
Acquisition of Kadima Neuropsychiatry Institute for CNS & Psychedelic Research Planned; New Drug Application for Suicidal Depression Treatment: $NRXP
TelAve News/10850235
NRx Pharmaceuticals, Inc.: Stock Symbol: NRXP IS Aiming to be the First FDA-Approved Medication to Treat Suicidal Depression
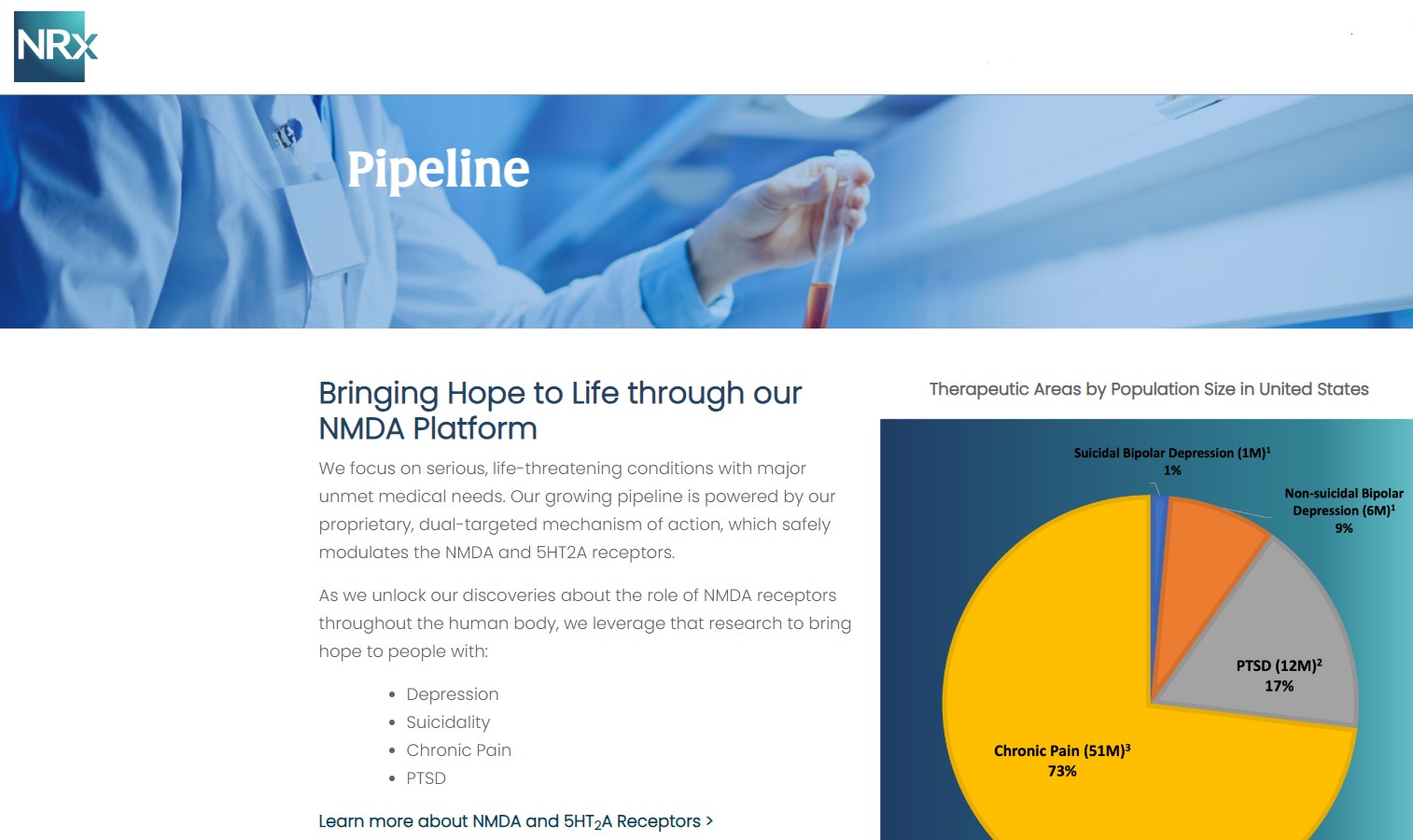
MIAMI - TelAve -- Developing NRX-101, an FDA-Designated Investigational Breakthrough Therapy for Suicidal Treatment-Resistant Bipolar Depression and Chronic Pain.
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
Kadima Neuropsychiatry Institute Targeted as First Acquisition for HOPE Subsidiary International Network of Interventional Psychiatry Clinics.
Kadima is a Leading Investigative Site for CNS and Psychedelic Research.
Completion of NDA Filing Expected in First Quarter of 2025
Plans to Participate in J.P. Morgan Healthcare Conference on January 13-16, 2025, in San Francisco, CA.
NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. $NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year. This represents a $3-5 billion market at expected pricing. Based on the data in the trials referenced above, the Company's regulatory counsel encouraged the Company to file an NDA for suicidal depression for NRX-100.
Kadima Neuropsychiatry Institute Expected First Acquisition for HOPE Subsidiary International Network of Interventional Psychiatry Clinics
More on TelAve News
On January 2nd NRXP announced the planned acquisition of the Kadima Neuropsychiatry Institute of La Jolla, CA, per the previously announced Letter of Intent, for the Company's HOPE subsidiary network. Kadima is expected to serve as the flagship clinic for HOPE's planned international network of interventional psychiatry clinics, designed to provide advanced treatments for debilitating diseases such as depression, anxiety and PTSD.
Kadima is one of the world's premier interventional psychiatry clinics and was among the first to introduce Ketamine Therapy for Central Nervous System (CNS) disorders at scale in the clinic setting. The clinic offers a full range of cutting-edge treatments for suicidal depression, anxiety, Post Traumatic Stress Disorder (PTSD) and other CNS disorders. Those treatment options include Ketamine Therapy, Spravato® (nasal esketamine), Transcranial Magnetic Stimulation as well as medication management.
Kadima also has a robust research division and is a leading investigative site for innovative CNS treatments, specializing in psychedelic research, for which it has served as a leading site in nearly all major clinical trials in this area. Kadima has contracts in place with the US Department of Veterans Affairs and also treats active-duty military personnel in the US Department of Defense under Tricare and other treatment programs.
Kadima's founder and CEO, Prof. David Feifel, MD PHD, has been a pioneer and international thought leader for advanced interventional treatment of psychiatric disorders such as depression, anxiety, PTSD and related disorders for more than three decades. Among other things, he is a co-author on a recent landmark expert consensus paper for treating depression with TMS, endorsed by three leading organizations in the field (ref). He also established the first clinical program to use subanesthetic dose ketamine infusions for neuropsychiatric disorders at UC San Diego, where he is currently Professor Emeritus of Psychiatry. His 150 peer-reviewed publications and several patents have provided global thought leadership on advanced approaches to treating psychiatric conditions, including integration of medicines like ketamine with neuromodulation such as, Transcranial Magnetic Stimulation and Digital Therapeutics. Kadima's experience will guide the growing network of NRXP HOPE Therapeutics clinics to an integrated, multi-modal approach to treating suicidal depression, anxiety and PTSD that is far more effective than ketamine alone.
More on TelAve News
Upon consummation of the proposed acquisition, Dr. Feifel will serve as NRXP HOPE's Chief Medical Innovation Officer (CMIO), focused on identifying and evaluating new developments in the treatment of CNS disorders and insuring Hope clinics are at the forefront of interventional psychiatry delivery, and leading global clinical trials to continue to advance the ability to treat these lethal diseases.
Dr. Feifel will join NRXP Chairman, Prof. Jonathan Javitt in presenting a Keynote address at the 8th Annual Neuroscience Innovation Forum during the JP Morgan Healthcare Conference in San Francisco, CA on the 12th of January 2025, and will join in meeting investors over the following week.
Initial Section of U.S. New Drug Application to the FDA for NRX-100 (IV Ketamine) for the Treatment of Suicidal Depression
On December 30th NRXP announced the transmission of first section of its New Drug Application (NDA) for NRX-100 (ketamine) for electronic filing with the U.S. Food & Drug Administration (FDA). NRX-100 was initially granted Fast Track Designation in 2017 for use in combination with NRX-101 (D-cycloserine/lurasidone) for treatment of suicidal bipolar depression. The Company is now seeking to expand the indication to include Suicidal Ideation in Major Depressive Disorder and other forms of depression, based on data from NIH- and European Government-funded trials that have been summarized on the NRXP website.
While assembly of the clinical data sections is being completed, FDA has asked the Company to submit the 1800-page manufacturing section (Module 3) of the NDA to enable immediate review prior to submission of final efficacy data and other sections of the NDA expected in the first quarter of 2025.
The NRx presentation of ketamine differs from the form of ketamine used in anesthesia in that it contains no potentially toxic preservatives and utilizes diversion-resistant packaging to enhance the traceability of a medicine known to have abuse potential.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
Disclaimer and Disclosure: https://corporateads.com/disclaimer/
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
Kadima Neuropsychiatry Institute Targeted as First Acquisition for HOPE Subsidiary International Network of Interventional Psychiatry Clinics.
Kadima is a Leading Investigative Site for CNS and Psychedelic Research.
Completion of NDA Filing Expected in First Quarter of 2025
Plans to Participate in J.P. Morgan Healthcare Conference on January 13-16, 2025, in San Francisco, CA.
NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. $NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year. This represents a $3-5 billion market at expected pricing. Based on the data in the trials referenced above, the Company's regulatory counsel encouraged the Company to file an NDA for suicidal depression for NRX-100.
Kadima Neuropsychiatry Institute Expected First Acquisition for HOPE Subsidiary International Network of Interventional Psychiatry Clinics
More on TelAve News
- Blackfoot Communications Expands Into New Rural Digital Opportunity Fund With netElastic vBNG and CGNAT Networking Software
- EFA Announces 2026 Editorial Rate Chart
- NYC Composer/Educator Launches Debut Children's Book to Fantastic Reviews
- Red5 Taps PubNub to Power the Next Era of Real-Time Interactive Streaming
- Shoutout Joseph Neibich aka Nybyk
On January 2nd NRXP announced the planned acquisition of the Kadima Neuropsychiatry Institute of La Jolla, CA, per the previously announced Letter of Intent, for the Company's HOPE subsidiary network. Kadima is expected to serve as the flagship clinic for HOPE's planned international network of interventional psychiatry clinics, designed to provide advanced treatments for debilitating diseases such as depression, anxiety and PTSD.
Kadima is one of the world's premier interventional psychiatry clinics and was among the first to introduce Ketamine Therapy for Central Nervous System (CNS) disorders at scale in the clinic setting. The clinic offers a full range of cutting-edge treatments for suicidal depression, anxiety, Post Traumatic Stress Disorder (PTSD) and other CNS disorders. Those treatment options include Ketamine Therapy, Spravato® (nasal esketamine), Transcranial Magnetic Stimulation as well as medication management.
Kadima also has a robust research division and is a leading investigative site for innovative CNS treatments, specializing in psychedelic research, for which it has served as a leading site in nearly all major clinical trials in this area. Kadima has contracts in place with the US Department of Veterans Affairs and also treats active-duty military personnel in the US Department of Defense under Tricare and other treatment programs.
Kadima's founder and CEO, Prof. David Feifel, MD PHD, has been a pioneer and international thought leader for advanced interventional treatment of psychiatric disorders such as depression, anxiety, PTSD and related disorders for more than three decades. Among other things, he is a co-author on a recent landmark expert consensus paper for treating depression with TMS, endorsed by three leading organizations in the field (ref). He also established the first clinical program to use subanesthetic dose ketamine infusions for neuropsychiatric disorders at UC San Diego, where he is currently Professor Emeritus of Psychiatry. His 150 peer-reviewed publications and several patents have provided global thought leadership on advanced approaches to treating psychiatric conditions, including integration of medicines like ketamine with neuromodulation such as, Transcranial Magnetic Stimulation and Digital Therapeutics. Kadima's experience will guide the growing network of NRXP HOPE Therapeutics clinics to an integrated, multi-modal approach to treating suicidal depression, anxiety and PTSD that is far more effective than ketamine alone.
More on TelAve News
- Meet Joseph Neibich aka Joseph Nybyk of Beachwood Canyon
- LARUS Launches Business Continuity Framework for IPv4-Dependent Networks
- KeysCaribbean Offers 'Skip-the-Crowds' Savings With 15 Percent Off April Stays
- Supply & Demand Chain Executive Names Puga Sankara as Recipient of 2026 Pros to Know Award
- AI Disruption Meets Marine Scale: Off The Hook YS, Inc. (N Y S E American: OTH) Targets Breakout Growth with NextBoat Launch and Aggressive Expansion
Upon consummation of the proposed acquisition, Dr. Feifel will serve as NRXP HOPE's Chief Medical Innovation Officer (CMIO), focused on identifying and evaluating new developments in the treatment of CNS disorders and insuring Hope clinics are at the forefront of interventional psychiatry delivery, and leading global clinical trials to continue to advance the ability to treat these lethal diseases.
Dr. Feifel will join NRXP Chairman, Prof. Jonathan Javitt in presenting a Keynote address at the 8th Annual Neuroscience Innovation Forum during the JP Morgan Healthcare Conference in San Francisco, CA on the 12th of January 2025, and will join in meeting investors over the following week.
Initial Section of U.S. New Drug Application to the FDA for NRX-100 (IV Ketamine) for the Treatment of Suicidal Depression
On December 30th NRXP announced the transmission of first section of its New Drug Application (NDA) for NRX-100 (ketamine) for electronic filing with the U.S. Food & Drug Administration (FDA). NRX-100 was initially granted Fast Track Designation in 2017 for use in combination with NRX-101 (D-cycloserine/lurasidone) for treatment of suicidal bipolar depression. The Company is now seeking to expand the indication to include Suicidal Ideation in Major Depressive Disorder and other forms of depression, based on data from NIH- and European Government-funded trials that have been summarized on the NRXP website.
While assembly of the clinical data sections is being completed, FDA has asked the Company to submit the 1800-page manufacturing section (Module 3) of the NDA to enable immediate review prior to submission of final efficacy data and other sections of the NDA expected in the first quarter of 2025.
The NRx presentation of ketamine differs from the form of ketamine used in anesthesia in that it contains no potentially toxic preservatives and utilizes diversion-resistant packaging to enhance the traceability of a medicine known to have abuse potential.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
Disclaimer and Disclosure: https://corporateads.com/disclaimer/
Source: Corporate Ads
0 Comments
Latest on TelAve News
- Tony Grundler Introduces Artificial Intelligence V.S. Avatar-Ian's
- Hollywood's Elite Gather at the Annual WOW Creations Oscars Gifting Suite at the Universal Hilton
- Where Were the Women? Reframing the Greek Revolution Through Contemporary Art
- JGCMGS Details Architecture to Safeguard Assets From Unauthorized Phishing Scams
- 21 Days: The Malta Deadline That Could Redraw the Finnish Online Casino Map
- JEGS Launches Modern, Secure Payments Powered by PhaseZero.ai
- U.S. Government Contracts in Excess of 38 Million Secured Through Partner, Establishing Multi-Year Defense Revenue Platform Through 2032: $BLIS
- New Report Reveals Surprising Trends in Ohio Airport Accidents
- Why Your Berberine Failed: RevGenetics Unveils the Absorption Gap Solution
- WCC Kitchens and Cabinets Featured on Selling Houses Australia
- Cat Hunt Simulator : Burrow & Pounce Lands on the App Store
- Shincheonji Marks 42nd Anniversary: From a Basement to a Denomination Growing by Tens of Thousands Annually
- Jackets for Jobs Hosts Smart & Sexy® Day Detroit for Women's History Month
- Tint Academy Training in Dallas Texas: Learn Window Tint & PPF Installation
- ASTPP Launches Advanced Telecom Billing Software to Simplify Billing for VoIP Providers
- $IBG accelerates toward transformative merger with BlockFuel as $6 million raise fuels dual-industry growth strategy: N A S D A Q: IBG
- High-Growth Power Infrastructure Play Targets AI Boom: 1606 Corp. Executes Aggressive Texas Expansion Strategy: 1606 Corp. (Stock Symbol: CBDW) $CBDW
- Accelerating the Transformation into a U.S. Nuclear Fuel Cycle Leader: Frontier Nuclear and Minerals Inc. (N A S D A Q: FNUC)
- Ozz Metals Ltd Secures 1-Tonne Gold Offtake Agreement
- Jet Set: The Ultimate Coachella Afterparty