Popular on TelAve
- Still Using Ice? FrostSkin Reinvents Hydration - 102
- Ice Melts. Infrastructure Fails. What Happens to Clean Water?
- Finland's €1.3 Billion Digital Gambling Market Faces Regulatory Tug-of-War as Player Protection Debate Intensifies
- Cold. Clean. Anywhere. Meet FrostSkin
- Amicly Launches as a Safety-First Social App Designed to Help People Build Real, Meaningful Friendships
- François Arnaud, star of Heated Rivalry, is the real-life inspiration behind Christopher Stoddard's novel At Night Only
- The Legal AI Showdown: Westlaw, Lexis, ChatGPT… or EvenSteven?
- Purple Heart Recipient Honored by Hall of Fame Son In Viral Tribute Sparking National Conversation on Service Fatherhood, Healing and Legacy
- Off The Hook YS (NY SE: OTH) Executes Transformational Apex Acquisition, Creating Vertically Integrated Marine Powerhouse with $60M Inventory Capacity
- From Sleepless Nights to Sold-Out Drops: Catch Phrase Poet's First Year Redefining Motivational Urban Apparel
Similar on TelAve
- Hypnotherapy Finder Announces Official US Wide Launch After Successful Soft Launch
- Melzi Job Coach Launches on iOS and Android: A Privacy-First AI Career Engine Built for Execution
- Training Lofts Launches $1,099 Unlimited Training Membership Featuring Semi-Private Coaching, Nutrition Support, and Recovery Services
- American Properties Realty, Inc. Leadership Attends NAHB International Builders' Show in Florida
- $317M Revenue and a Clear Path to $1B: $IQST is Positioned for a Major Profitability Inflection
- ASI Hosts 2026 Executive Business Summit for Global Partner Community
- $6 Million Funding Secured as Retail Expansion, Operational Streamlining, and Asset-Light Strategy Position the Company for Accelerated Growth $SOWG
- K2 Integrity Enhances Technology Capabilities Through Acquisition of Leviathan Security Group
- Buildout Launches CRM, Completing the Industry's First AI-Powered End-to-End Deal Engine for CRE
- Kanguro Insurance Taps Paylode to Launch Best-in-Class Pet and Renters Insurance Rewards Experience
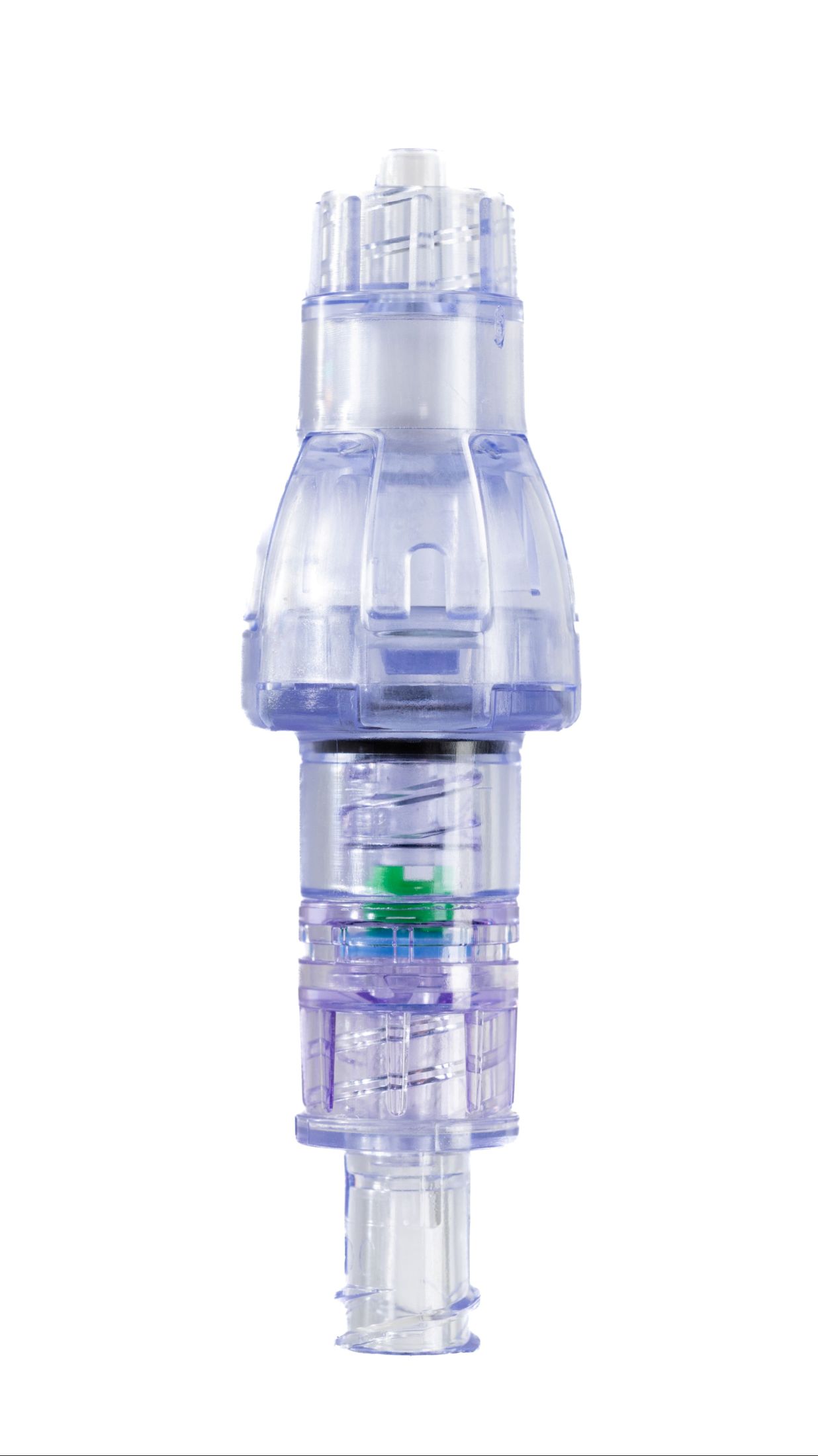
Lineus Medical Completes UK Registration for SafeBreak® Vascular
TelAve News/10884995
FAYETTEVILLE, Ark. - TelAve -- Lineus Medical is now officially registered in the United Kingdom, enabling the company to begin distributing SafeBreak® Vascular within the UK healthcare market. UK registration represents an important step in Lineus Medical's international expansion strategy, further extending access to SafeBreak Vascular for hospitals and clinicians on a global scale. With regulatory requirements in place, Lineus Medical can now work with distribution partners to bring its breakaway IV technology to the UK.
SafeBreak Vascular is a breakaway device for IV lines clinically proven to reduce IV complications.¹ When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When separation occurs, valves on both ends of the device close to prevent medication spills from the pump and blood loss from the patient. Patients avoid additional needlesticks, nurses save time, and hospitals save money.¹
More on TelAve News
"Completing UK registration is another meaningful milestone as we continue to expand access to SafeBreak globally," said Vance Clement, CEO of Lineus Medical. "Each new market brings us closer to our mission of removing the pains associated with IV lines and improving patient safety across healthcare systems worldwide. UK registration allows SafeBreak to reach another 69 million people."
"This registration allows us to move forward with distribution planning in the UK and supports our broader international commercialization efforts," said Larry Hayes, Chief Commercial Officer of Lineus Medical. "We are focused on working with the right partners to ensure SafeBreak Vascular is accessible to clinicians who are looking to reduce IV complications and improve care at the bedside."
About Lineus Medical:
Lineus Medical is the developer of SafeBreak® Vascular, a breakaway technology proven to reduce IV restarts and IV complications.1 Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn, Facebook, and Instagram.
References
SafeBreak Vascular is a breakaway device for IV lines clinically proven to reduce IV complications.¹ When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When separation occurs, valves on both ends of the device close to prevent medication spills from the pump and blood loss from the patient. Patients avoid additional needlesticks, nurses save time, and hospitals save money.¹
More on TelAve News
- ASI Hosts 2026 Executive Business Summit for Global Partner Community
- Pastor Saeed Abedini Releases THE TRUTH – Volume 1, A Deeply Personal Story of Faith, Struggle, and Redemption
- New Book Warring From the Standpoint of the Throne Room Calls Believers to Pray From Victory
- Scotch Whisky Market Dislocation Creates Compelling Entry Opportunity for Long-Term Investors
- Peccioli Becomes New Orleans: In July 2026, the magic of jazz comes to Tuscany
"Completing UK registration is another meaningful milestone as we continue to expand access to SafeBreak globally," said Vance Clement, CEO of Lineus Medical. "Each new market brings us closer to our mission of removing the pains associated with IV lines and improving patient safety across healthcare systems worldwide. UK registration allows SafeBreak to reach another 69 million people."
"This registration allows us to move forward with distribution planning in the UK and supports our broader international commercialization efforts," said Larry Hayes, Chief Commercial Officer of Lineus Medical. "We are focused on working with the right partners to ensure SafeBreak Vascular is accessible to clinicians who are looking to reduce IV complications and improve care at the bedside."
About Lineus Medical:
Lineus Medical is the developer of SafeBreak® Vascular, a breakaway technology proven to reduce IV restarts and IV complications.1 Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn, Facebook, and Instagram.
References
- Data on file.
Source: Lineus Medical
Filed Under: Technology
0 Comments
Latest on TelAve News
- Grads aren't getting hired — here's what we're doing about it
- K2 Integrity Enhances Technology Capabilities Through Acquisition of Leviathan Security Group
- #WeAreGreekWarriors Comes to Detroit in Celebration of Women's History Month
- Energywise Solutions and Pickleball Pros Partner to Bring More Energy and Visibility to Pickleball Clubs
- Buildout Launches CRM, Completing the Industry's First AI-Powered End-to-End Deal Engine for CRE
- The Franchise King® Releases Free Guide for Nervous Buyers
- Kanguro Insurance Taps Paylode to Launch Best-in-Class Pet and Renters Insurance Rewards Experience
- CCHR: CIA Mind-Control Files Raise Urgent Questions as Millions Take Psychotropic Drugs
- NRx Pharmaceuticals Launches Breakthrough One-Day Treatment Clinic in Florida as FDA Pathway and Clinical Data Strengthen Growth Outlook; $NRXP
- Revenue Optics Launches Talent Infrastructure Platform for SaaS Revenue Hiring and Appoints Sabz Kaur to Lead Growth
- Building a Multi-Domain Autonomous Systems Platform at the Intersection of AI, Defense and Infrastructure: VisionWave Holdings (N A S D A Q: VWAV)
- Bent Danholm Named "Top Luxury Real Estate Leader" in Modern Luxury Miami
- Author Ken Mora to Celebrate New Caravaggio Book Debut with Special Event at Palazzo Venezia Naples
- Matthew Sisneros Releases Raw and Unfiltered Memoir: The Devil Lost Another One — A Powerful Story of Crime, Consequence, and Redemption
- From Life to Light: Jess L. Martinez Shares a Soulful Poetry Collection That Explores What It Means to Be Human
- Lawsuit Filed Against Boeing Over Defective Seat Switch on Boeing 787
- Quadcode Acquires Significant Stake in Game 7, LLC - The Parent Company for FPFX Tech and PropAccount.com
- Danholm Collection Announces Sale of 16689 Broadwater Ave in Winter Garden, Highlighting Strong Performance in Twinwaters Community
- Strong Clinical Results for Breakthrough Liver Diagnostic Platform; ENDRA Life Sciences (N A S D A Q: NDRA) $NDRA
- 46th International Symposium On Forecasting – Dates, Venue And Speakers Announced